A common source of confusion is that many individuals with these symptoms are informed that their laboratory results are “within normal limits.”
Dr. Kalpana Gupta Shekhawat
MBBS, DNB, DFM
MD – Clinical Nutrition (CNCB, Texas, USA)
Functional & Regenerative Medicine Specialist
BHRT Consultant (American Academy of Anti-Aging Medicine)
Certified in Peptide Therapy (Peptide Therapy Foundation & A4M, USA)
Dr. Kalpana Gupta Shekhawat brings over 15 years of clinical experience, with focused expertise in autoimmune, metabolic, neurological, and chronic health conditions. Her work includes autoimmune gut disorders such as IBS and IBD; neurological conditions including multiple sclerosis, anxiety, depression, autism, and fibromyalgia; mast cell activation syndrome (MCAS); metabolic disorders such as Hashimoto’s thyroiditis and diabetes; hormonal imbalances; and latent infections.

Her clinical approach centers on identifying and addressing the root causes of disease through personalized, patient-focused care. She integrates nutrition, gut health optimization, detoxification, and advanced therapeutic interventions—including hormone balancing with bioidentical hormone replacement therapy (BHRT), peptide therapy, and IV protocols—to deliver evidence-based care aimed at restoring metabolic balance and supporting long-term health outcomes.
Nighttime restlessness in the legs is often oversimplified. Magnesium, stretching, or stress are commonly considered the primary causes, and while these factors may offer partial relief, they do not fully explain a pattern many individuals consistently report—an urge to move the legs that emerges predominantly at night, just as the body is attempting to transition into rest.
When symptoms follow this distinct timing, it becomes important to look beyond the muscles and consider the role of the central nervous system.
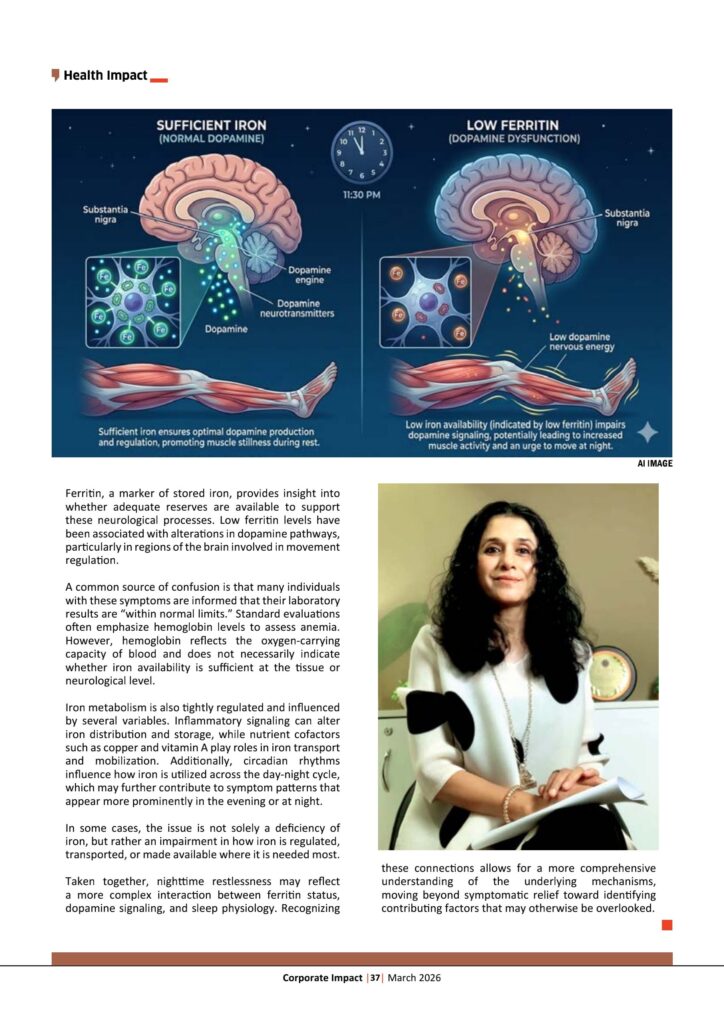
Within the midbrain lies the substantia nigra, a small but essential structure involved in the production of dopamine and the regulation of movement. Dopamine is widely recognized for its role in motor control during waking hours, but its influence extends into sleep physiology. Adequate dopamine signaling helps suppress unnecessary movement at night, allowing the body to remain still and progress into deeper, restorative stages of sleep. When this signaling is suboptimal, the ability to maintain restfulness may be compromised.
One key factor influencing this pathway is iron.
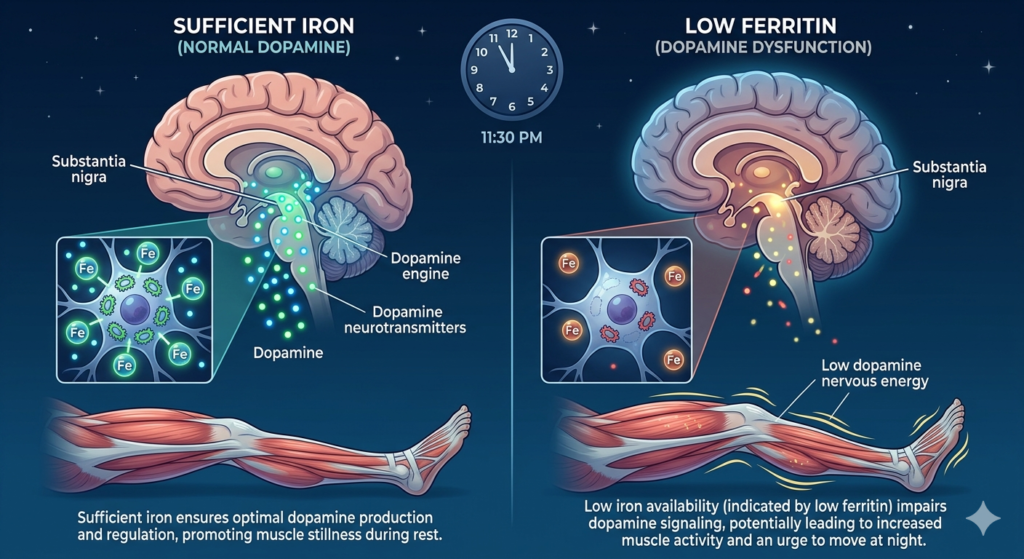
Iron is required for the activity of tyrosine hydroxylase, the enzyme responsible for converting tyrosine into L-DOPA—the first and rate-limiting step in dopamine synthesis. Without sufficient iron availability, this process becomes less efficient, potentially affecting both dopamine production and downstream signaling.
Ferritin, a marker of stored iron, provides insight into whether adequate reserves are available to support these neurological processes. Low ferritin levels have been associated with alterations in dopamine pathways, particularly in regions of the brain involved in movement regulation.
A common source of confusion is that many individuals with these symptoms are informed that their laboratory results are “within normal limits.” Standard evaluations often emphasize hemoglobin levels to assess anemia. However, hemoglobin reflects the oxygen-carrying capacity of blood and does not necessarily indicate whether iron availability is sufficient at the tissue or neurological level.
Iron metabolism is also tightly regulated and influenced by several variables. Inflammatory signaling can alter iron distribution and storage, while nutrient cofactors such as copper and vitamin A play roles in iron transport and mobilization. Additionally, circadian rhythms influence how iron is utilized across the day-night cycle, which may further contribute to symptom patterns that appear more prominently in the evening or at night.
In some cases, the issue is not solely a deficiency of iron, but rather an impairment in how iron is regulated, transported, or made available where it is needed most.
Taken together, nighttime restlessness may reflect a more complex interaction between ferritin status, dopamine signaling, and sleep physiology. Recognizing these connections allows for a more comprehensive understanding of the underlying mechanisms, moving beyond symptomatic relief toward identifying contributing factors that may otherwise be overlooked.